Protein-coding gene in the species Homo sapiens
| PLS3 |
|---|
 |
| Available structures |
|---|
| PDB | Ortholog search: PDBe RCSB |
|---|
| List of PDB id codes |
|---|
1AOA, 1WJO |
|
|
| Identifiers |
|---|
| Aliases | PLS3, BMND18, T-plastin, plastin 3 |
|---|
| External IDs | OMIM: 300131; MGI: 104807; HomoloGene: 128200; GeneCards: PLS3; OMA:PLS3 - orthologs |
|---|
| Gene location (Human) |
|---|
 | | Chr. | X chromosome (human)[1] |
|---|
| | Band | Xq23 | Start | 115,561,174 bp[1] |
|---|
| End | 115,650,861 bp[1] |
|---|
|
| Gene location (Mouse) |
|---|
 | | Chr. | X chromosome (mouse)[2] |
|---|
| | Band | X|X A7.3 | Start | 74,829,260 bp[2] |
|---|
| End | 74,918,788 bp[2] |
|---|
|
| RNA expression pattern |
|---|
| Bgee | | Human | Mouse (ortholog) |
|---|
| Top expressed in | - Achilles tendon
- visceral pleura
- parietal pleura
- skin of hip
- amniotic fluid
- Descending thoracic aorta
- ascending aorta
- right coronary artery
- oral cavity
- skin of thigh
|
| | Top expressed in | - tunica media of zone of aorta
- subiculum
- ascending aorta
- conjunctival fornix
- efferent ductule
- pontine nuclei
- belly cord
- carotid body
- molar
- mammillary body
|
| | More reference expression data |
|
|---|
| BioGPS | 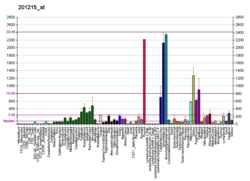 | | More reference expression data |
|
|---|
|
| Gene ontology |
|---|
| Molecular function | - calcium ion binding
- actin binding
- metal ion binding
- actin filament binding
| | Cellular component | - cytoplasm
- cytosol
- actin filament
- plasma membrane
- actin filament bundle
| | Biological process | - bone development
- actin filament bundle assembly
- actin filament network formation
- actin crosslink formation
| | Sources:Amigo / QuickGO |
|
| Orthologs |
|---|
| Species | Human | Mouse |
|---|
| Entrez | | |
|---|
| Ensembl | | |
|---|
| UniProt | | |
|---|
| RefSeq (mRNA) | |
|---|
NM_001136025
NM_001172335
NM_001282337
NM_001282338
NM_005032 |
| |
|---|
NM_001166453
NM_001166454
NM_145629
NM_001346519
NM_001346520 |
|
|---|
| RefSeq (protein) | |
|---|
NP_001129497
NP_001165806
NP_001269266
NP_001269267
NP_005023 |
| |
|---|
NP_001159925
NP_001159926
NP_001333448
NP_001333449
NP_663604 |
|
|---|
| Location (UCSC) | Chr X: 115.56 – 115.65 Mb | Chr X: 74.83 – 74.92 Mb |
|---|
| PubMed search | [3] | [4] |
|---|
|
| Wikidata |
| View/Edit Human | View/Edit Mouse |
|
Plastin-3 is a highly conserved protein that in humans is encoded by the PLS3 gene on the X chromosome.[5][6]
Function
Plastins are a family of actin-binding proteins that are conserved throughout eukaryote evolution and expressed in most tissues of higher eukaryotes. In humans, two ubiquitous plastin isoforms (L and T) have been identified. Plastin 1 (otherwise known as Fimbrin) is a third distinct plastin isoform which is specifically expressed at high levels in the small intestine. The L isoform is expressed only in hemopoietic cell lineages, while the T isoform has been found in all other normal cells of solid tissues that have replicative potential (fibroblasts, endothelial cells, epithelial cells, melanocytes, etc.). The C-terminal 570 amino acids of the T-plastin and L-plastin proteins are 83% identical. It contains a potential calcium-binding site near the N-terminus.[6]
Clinical significance
Defects in PLS3 are associated with osteoporosis and bone fracture in humans and in knockout zebrafish.[7]
References
- ^ a b c GRCh38: Ensembl release 89: ENSG00000102024 – Ensembl, May 2017
- ^ a b c GRCm38: Ensembl release 89: ENSMUSG00000016382 – Ensembl, May 2017
- ^ "Human PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ "Mouse PubMed Reference:". National Center for Biotechnology Information, U.S. National Library of Medicine.
- ^ Lin CS, Park T, Chen ZP, Leavitt J (Mar 1993). "Human plastin genes. Comparative gene structure, chromosome location, and differential expression in normal and neoplastic cells". J Biol Chem. 268 (4): 2781–92. doi:10.1016/S0021-9258(18)53842-4. PMID 8428952.
- ^ a b "Entrez Gene: PLS3 plastin 3 (T isoform)".
- ^ van Dijk FS, Zillikens MC, Micha D, Riessland M, Marcelis CL, de Die-Smulders CE, Milbradt J, Franken AA, Harsevoort AJ, Lichtenbelt KD, et al. (October 2013). "PLS3 Mutations in X-Linked Osteoporosis with Fractures". N. Engl. J. Med. 369 (16): 1529–36. CiteSeerX 10.1.1.713.901. doi:10.1056/NEJMoa1308223. PMID 24088043.
Further reading
- Lin CS, Aebersold RH, Leavitt J (1990). "Correction of the N-terminal sequences of the human plastin isoforms by using anchored polymerase chain reaction: identification of a potential calcium-binding domain". Mol. Cell. Biol. 10 (4): 1818–21. doi:10.1128/MCB.10.4.1818. PMC 362293. PMID 2378651.
- Lin CS, Aebersold RH, Kent SB, et al. (1988). "Molecular cloning and characterization of plastin, a human leukocyte protein expressed in transformed human fibroblasts". Mol. Cell. Biol. 8 (11): 4659–68. doi:10.1128/MCB.8.11.4659. PMC 365555. PMID 3211125.
- Goldstein D, Djeu J, Latter G, et al. (1985). "Abundant synthesis of the transformation-induced protein of neoplastic human fibroblasts, plastin, in normal lymphocytes". Cancer Res. 45 (11 Pt 2): 5643–7. PMID 4053036.
- Arpin M, Friederich E, Algrain M, et al. (1995). "Functional differences between L- and T-plastin isoforms". J. Cell Biol. 127 (6 Pt 2): 1995–2008. doi:10.1083/jcb.127.6.1995. PMC 2120298. PMID 7806577.
- Maruyama K, Sugano S (1994). "Oligo-capping: a simple method to replace the cap structure of eukaryotic mRNAs with oligoribonucleotides". Gene. 138 (1–2): 171–4. doi:10.1016/0378-1119(94)90802-8. PMID 8125298.
- Lin CS, Shen W, Chen ZP, et al. (1994). "Identification of I-plastin, a human fimbrin isoform expressed in intestine and kidney". Mol. Cell. Biol. 14 (4): 2457–67. doi:10.1128/mcb.14.4.2457. PMC 358613. PMID 8139549.
- Goldsmith SC, Pokala N, Shen W, et al. (1997). "The structure of an actin-crosslinking domain from human fimbrin". Nat. Struct. Biol. 4 (9): 708–12. doi:10.1038/nsb0997-708. PMID 9302997. S2CID 9506514.
- Shoeman RL, Hartig R, Hauses C, Traub P (2003). "Organization of focal adhesion plaques is disrupted by action of the HIV-1 protease". Cell Biol. Int. 26 (6): 529–39. doi:10.1006/cbir.2002.0895. PMID 12119179. S2CID 39778155.
- Rao RM, Rama S, Rao AJ (2004). "Changes in T-plastin expression with human trophoblast differentiation". Reprod. Biomed. Online. 7 (2): 235–42. doi:10.1016/S1472-6483(10)61758-0. PMID 14567899.
- Su MW, Dorocicz I, Dragowska WH, et al. (2004). "Aberrant expression of T-plastin in Sezary cells". Cancer Res. 63 (21): 7122–7. PMID 14612505.
- Giganti A, Plastino J, Janji B, et al. (2005). "Actin-filament cross-linking protein T-plastin increases Arp2/3-mediated actin-based movement". J. Cell Sci. 118 (Pt 6): 1255–65. doi:10.1242/jcs.01698. PMID 15741236.
- Ralser M, Nonhoff U, Albrecht M, et al. (2005). "Ataxin-2 and huntingtin interact with endophilin-A complexes to function in plastin-associated pathways". Hum. Mol. Genet. 14 (19): 2893–909. doi:10.1093/hmg/ddi321. hdl:11858/00-001M-0000-0010-85BB-5. PMID 16115810.
- Ikeda H, Sasaki Y, Kobayashi T, et al. (2006). "The role of T-fimbrin in the response to DNA damage: silencing of T-fimbrin by small interfering RNA sensitizes human liver cancer cells to DNA-damaging agents". Int. J. Oncol. 27 (4): 933–40. doi:10.3892/ijo.27.4.933. PMID 16142308.
 | This article on a gene on the human X chromosome and/or its associated protein is a stub. You can help Wikipedia by expanding it. |

 1aoa: N-Terminal Actin-Crosslinking domain from human fimbrin
1aoa: N-Terminal Actin-Crosslinking domain from human fimbrin 1wjo: Solution structure of the fourth CH domain from human plastin 3 T-isoform
1wjo: Solution structure of the fourth CH domain from human plastin 3 T-isoform



















